

SILANAT
1st inorganic Si3N4-based fuel
Prologue:
©SILANAT is a new energy carrier based on silicon (from sand) and nitrogen (from the air). The first inorganic fuel in the world, ©SILANAT burns ad absolutum without CO2 and leaves no toxins behind. In other words: ©SILANAT reacts thermally with air (≈ 20 % oxygen and ≈ 80 % nitrogen) without producing CO2 or other pollutants. The ©SILANAT off-gas then consists of water, silicon nitride and nitrates – all non-toxic and residue-free. How is this possible? Because it is a logical consequence of the fact that fossil fuels are based on hydrocarbons (CmHn), while ©SILANAT consists of hydrosilanes (SinH2n+2). This is significant in that all reciprocating internal combustion engines and / or jet engines used today heat up the nitrogen present in the atmosphere by almost 80 % due to CO2 emissions, thus causing global warming in the first place. A natural fact about whose causation the general literature is conspicuously silent. ©SILANAT, on the other hand, cools these huge amounts of heated atmospheric nitrogen by burning nitrogen, which at the same time substantially increases its efficiency. Ergo, with ©SILANAT, much of the combustion energy is not released to the atmosphere or lost as climate/environmentally damaging heat energy, but is used for actual engine power, resulting in >90% efficiency (compared to fossil fuels at ≈ 30%). Consider the somewhat limp comparison: LED versus burning hot light bulb!

Art. 1
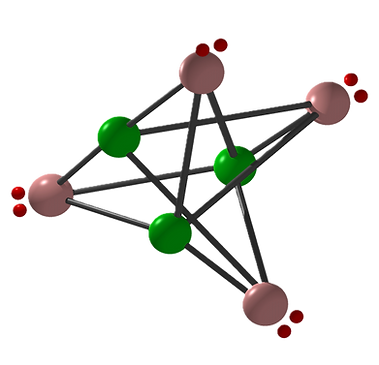
What is Si3N4?
The 3 silicon atoms (green) each have 4 free valences, represented by (black) bond arms. Each of the 4 pentavalent nitrogen atoms (reddish) has 3 bond arms to the silicon atom and 1 free electron pair (small red colons). In realty, however, Si3N4 consists of 4 ∙ 3 nitrogen-silicon bonds and 3 ∙ 4 silicon-nitrogen bonds, i.e. a total of 24 valence electrons plus 8 (4 ∙ 2) tetrahedrally arranged electrons on the outside.
Si3N4 thus fulfills the decisive criteria of a noble gas such as neon, argon, krypton, xenon… And since each molecular unit retains its 4 outwardly directed electron pairs even in the solid state, one speaks of a "fossilized noble gas", which means that Si3N4 is considered to be completely non-toxic. Furthermore, it should be noted that the fact that there are 3 silicon atoms for every 4 nitrogen atoms in this molecule (Si3N4 thus consists of a total of 7 atoms) suggests that it is a type of implosion explosive due to this controllable prime number arrangement. Based on this finding, silanes can be burned in the form of a liquid inorganic fuel with pent-up hot air, so that the ≈ 20 % oxygen in the air can only burn to silicon, while the ≈ 80% nitrogen in the air burns the excess silicon atoms to Si3N4.
Art. 2
What is ©SILANAT?
The introduction of ©SILANAT as a new drive and fuel system means, first of all, that the raw material required for it is much cheaper than in the production of conventional fuels thanks to a new chemical process. At the same time, the environment and the climate are substantially relieved in this process. The basic raw materials are sand and nitrogen. They are first used to interproduce an oil-tar mixture, which is then split into carbon and hydrogen, while the sands are converted into gaseous silicon fluoride. The latter, in turn, is converted into crystalline silicon with the help of aluminum granules (also thermally). The silicon obtained in this way is extremely inexpensive and of highest quality, even by today's standards. The “interproduced” oil-tar mixture is itself a solid hydrocarbon that releases hydrogen at high heat. This in turn regulates pyrolysis. The resulting excess gas is converted into three-phase electricity or fed into natural gas grids, which in the case of ©SILANAT production also opens up new climate-friendly source of energy in general. Last but not least, silicon, a chemical element similar to carbon, doesn’t produce gaseous dioxide, but burns with the nitrogen in the air to form silicon nitrogen (Si3N4). This type of combustion produces no hot gases other than clear water vapor. So, the efficiency of ©SILANAT is over 90 %. This is partly due to the fact that the atmosphere is not heated during the combustion process.

By comparison, the efficiency of burning fossil fuels such as natural gas, coal, oil, etc. is between 10 – 30 %. This low efficiency is due, on the one hand, to the fact that carbon-containing compounds react with the oxygen in the air and the resulting hot CO2 also heats up the nitrogen, which makes up the largest part of the earth's atmosphere, and, on the other hand, the energy calculation formula used here contains incorrect formula variables. (I deal with the last point separately). In any case, combustion with fossil fuels always requires a chimney or exhaust system that unnecessarily heats up the earth's atmosphere. To escape this trap, there is now “©SILANAT”, which namely contains no carbon at all, especially since it reacts exclusively with nitrogen in the air… Additional advantages:

Art. 3
Basic consideration
A note upfront: Most fuels of our modern industrial age are based on the combustion of oxygen. The most common "waste gas" is ultimately burnt carbon (CO2). Whether the latter is actually considered a major climate killer or should rather be weighted as an atmospherically scarce resource, since carbon is sometimes the most important building material for our already dying (starving) vegetation, I leave open, as such an assessment is not the purpose of this short essay. Oxygen itself is the most abundant element on earth, although it makes up only ≈ 20 % of the earth's atmosphere (CO2 ≈ 0.04 %). Nitrogen, on the other hand, occurs predominantly in gaseous form and makes up nearly 80 % of our atmosphere. Finally we come to silicon, the second most abundant element on earth. The following can therefore be anticipated with regard to ©SILANAT: Since we are dealing with a new combustible fuel, its gaseous resource is crucial. And since silicon, along with oxygen, makes up the largest proportion of the earth's crust (≈ 25 %), it could hardly be better suited as a basic raw material and new type of climate-friendly fuel, together with nitrogen (≈ 80 % of the earth's atmosphere) as an aid to combustion.

Art. 4
Fundamental specification
Above all, however, silicon -in contrast to carbon- has the special property of forming a highly stable nitride compound, which in the case of the silicon(tetra)nitride compound (Si3N4) is, inter alia, possible by the action of molecular nitrogen on silicon powder. The temperature required for this, subject to the otherwise correctly applied parameters, is approx. 90°C. Normally, up to 3,000°C is required for such a compound under the influence of N, which can then, however, also lead to bomb-like explosions. Already in the course of this low-temperature production lies a far-reaching USP for our new fuel. For the production of Si3N4, desert sand (SiO2) can be used as the raw material source from which to obtain pure silicon in the first and simplest step. (Compare the respective attached patent specifications).
Art. 5
Manufacturing
Higher silanes from this pure silicon are now prepared (such as under repeated modified Mueller-Rochow synthesis with silyl chlorides), whereby those silyl chlorides could either be silicone chemical waste or could be obtained from mono- and/or disilane. The higher silanes thus added are subsequently burned -releasing energy- with air to form water and silicon nitride (Si3N4). The silicon nitride is converted into ammonia (NH3) with the formation of silicates, in order to be split into nitrogen + water in the further course of combustion, thus closing the nitrogen cycle. Silicon (Si) is located just below carbon (C) in the periodic table of chemical elements and is consequently very similar to the latter. However, the hydrogen compounds of silicon have some differences to hydrocarbons. Thus, it should be primarily considered that di-/tri-/tetrasilane are self-igniting in air. This makes it all the more important to use higher silanes, such as penta-/decasilane, the producibility of which was proven as early as 1968 at a Cologne university. The critical factor is that higher silanes become increasingly stable with increasing chain length, so that heptasilane, for example, is no longer self-igniting from room temperature, making higher silanes much safer to handle, "less toxic" and, above all, – good as – diesel identical.
Art. 6
Nature illustration
As a final remark on the producibility and applicability of ©SILANAT, the following comparison from nature is given. A phenomenon that everyone is familiar with: The air is sticky and the sky eerie. Dark squadrons of clouds gather above us – silence! First raindrops. Thunder. The battle begins. … Glistening, billion-volt lightning discharges transform the atmosphere into an awe-inspiring spectacle of hell. This happens several times a year. Thank God. Because without these kinds of thunderstorms, life on earth would not be possible. The reason: the plants need a constant supply of nitric nitrogen fertilizer for the formation of their (19 +1) amino acids. They get the nitrogen they need for this from the air, because otherwise there will be no nitrates in the soil. Hence the need for lightning:

The nitrogen bound up in the atmosphere opens its triple bond when there are enough static electrons and reacts (with the aid of the energy in the lightning) with oxygen to form nitrogen oxides. The thunderstorm washes out the resulting nitric acid and thus transports the nitrogen compounds into the soil. The plants are thus regularly supplied with nitrogen and are therefore able to produce amino acids in the first place. However, this natural miracle is only possible because the unique triple bond of the nitrogen molecule remains stable against all other chemical influences. It must be said that there are only 3 elements in total that can form single, double and triple bonds: Oxygen, Carbon and Nitrogen. With regard to silicon, the latter can only react with nitrogen only singly, since silicon itself cannot form a triple bond. Consequently: Silicon burns with nitrogen! Thank God there is no silicon in the atmosphere, because otherwise the world would "blow up" during a thunderstorm. The unimaginable lightning discharges are thus the result of an otherwise impossible opening of that unique, stable triple bond of nitrogen in the air. The forces behind this event are so enormous that it is still not possible to measure them in reality or even to use them. This release of energy already gives an idea of the tremendous power that is concentrated in nitrogen.
Art. 7
Final conclusion
Silicon hydrogens in the form of silane oil react with air in the combustion chamber of a ©SILANAT-powered engine, initially generating increased temperatures. The nitrogen in the air flowing in (content in the air is approx. 80 %) then reacts with silicon in the silicon hydride, which results in the formation of silicon (tetra) nitride (Si3N4). The rest is then split into water + ammonia, the former being transported into the soil by the latter, similar to thunderstorms, where the plants are constantly waiting for nitrates to form their (19 + 1) amino acids. A first and unique win-win situation for energy generation and environmental support at the same time! Finally, as far as the silicon nitride is concerned, this is a “petrified noble gas” the hardness of which is approximately equal to that of a diamond. The industrial benefits of this waste are obvious. In conclusion: ©SILANAT has three USPs:
-
Optimal energy value creation;
-
Environmental relief and environmental aid;
-
A by-product for industry as a diamond substitute.